Atoms And Ions Worksheet Answer Key
Onlines
Apr 03, 2025 · 7 min read
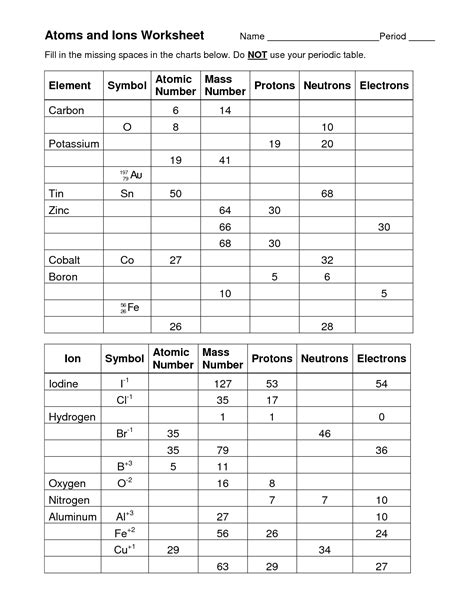
Table of Contents
Atoms and Ions Worksheet Answer Key: A Comprehensive Guide
Understanding atoms and ions is fundamental to grasping the basics of chemistry. This comprehensive guide provides answers and explanations to common questions found in atoms and ions worksheets. We'll cover key concepts like atomic structure, ionic bonding, and the differences between atoms and ions, all while applying SEO best practices for improved searchability. This guide aims to be your ultimate resource for mastering this crucial area of chemistry.
Atomic Structure: The Building Blocks of Matter
Atoms, the fundamental units of matter, are composed of three subatomic particles:
- Protons: Positively charged particles located in the atom's nucleus. The number of protons defines the element's atomic number and determines its identity.
- Neutrons: Neutrally charged particles also residing in the nucleus. Neutrons contribute to the atom's mass but not its charge.
- Electrons: Negatively charged particles orbiting the nucleus in energy levels or shells. The number of electrons generally equals the number of protons in a neutral atom.
Isotopes and Atomic Mass
Isotopes are atoms of the same element with the same number of protons but a different number of neutrons. This variation in neutron number leads to different atomic masses for the isotopes of an element. The atomic mass (or atomic weight) is the weighted average mass of all naturally occurring isotopes of an element. It's crucial to understand that atomic mass is not a whole number because it reflects the abundance of different isotopes.
Example: Carbon-12 and Carbon-14 are isotopes of carbon. Both have 6 protons, but Carbon-12 has 6 neutrons while Carbon-14 has 8 neutrons. The atomic mass of carbon is approximately 12.01 amu, reflecting the abundance of Carbon-12.
Electron Configuration and Valence Electrons
Electrons occupy specific energy levels or shells around the nucleus. The arrangement of electrons in these shells is called the electron configuration. Valence electrons are the electrons in the outermost shell. These electrons are crucial in determining an atom's chemical reactivity and how it will bond with other atoms. The number of valence electrons often dictates the number of bonds an atom can form.
Example: Oxygen (atomic number 8) has an electron configuration of 2, 6. It has six valence electrons, which means it can form two covalent bonds to achieve a stable octet (eight electrons in its outermost shell).
Ions: Charged Particles
An ion is an atom or molecule that has gained or lost one or more electrons, resulting in a net electrical charge. Ions are formed through the process of ionization.
- Cations: Positively charged ions formed when an atom loses one or more electrons. Metals tend to form cations.
- Anions: Negatively charged ions formed when an atom gains one or more electrons. Nonmetals tend to form anions.
Ionic Bonding: The Attraction of Opposites
Ionic bonding is the electrostatic attraction between oppositely charged ions. This type of bond typically occurs between a metal and a nonmetal. The metal atom loses electrons to become a cation, and the nonmetal atom gains these electrons to become an anion. The resulting electrostatic attraction holds the ions together in a crystal lattice structure.
Example: Sodium (Na) readily loses one electron to become Na⁺ (sodium cation), while chlorine (Cl) readily gains one electron to become Cl⁻ (chloride anion). The electrostatic attraction between Na⁺ and Cl⁻ forms sodium chloride (NaCl), or table salt.
Worksheet Questions and Answers (Illustrative Examples)
The following sections provide example questions and answers commonly found in atoms and ions worksheets. Remember, specific questions and answers will vary based on the worksheet's content and difficulty level.
Question 1: Identifying Protons, Neutrons, and Electrons
Question: An atom of oxygen has an atomic number of 8 and an atomic mass of 16. How many protons, neutrons, and electrons are present in a neutral oxygen atom?
Answer:
- Protons: The atomic number (8) represents the number of protons.
- Neutrons: Atomic mass (16) – Atomic number (8) = 8 neutrons.
- Electrons: In a neutral atom, the number of electrons equals the number of protons (8).
Question 2: Determining the Charge of Ions
Question: Magnesium (Mg) typically loses two electrons to form an ion. What is the charge of the magnesium ion?
Answer: Losing two negatively charged electrons leaves the magnesium ion with a net charge of +2 (Mg²⁺).
Question 3: Writing Ionic Formulas
Question: Write the formula for the ionic compound formed between calcium (Ca) and chlorine (Cl).
Answer: Calcium (Ca) forms a +2 ion (Ca²⁺), and chlorine (Cl) forms a -1 ion (Cl⁻). To balance the charges, two chlorine ions are needed for each calcium ion. Therefore, the formula is CaCl₂.
Question 4: Electron Configuration and Valence Electrons
Question: What is the electron configuration of nitrogen (N), and how many valence electrons does it have?
Answer: Nitrogen (atomic number 7) has an electron configuration of 2, 5. It has five valence electrons.
Question 5: Isotopes and Atomic Mass
Question: Explain the concept of isotopes, giving an example.
Answer: Isotopes are atoms of the same element with the same number of protons but a different number of neutrons. This means they have the same atomic number but different atomic masses. For example, carbon-12 (⁶C¹²) and carbon-14 (⁶C¹⁴) are isotopes of carbon. Both have 6 protons, but carbon-12 has 6 neutrons, while carbon-14 has 8 neutrons.
Question 6: Ionic vs. Covalent Bonding
Question: What is the key difference between ionic and covalent bonding? Give examples of each.
Answer: Ionic bonding involves the transfer of electrons between atoms, creating ions that are held together by electrostatic attraction. This typically occurs between a metal and a nonmetal. Example: NaCl (sodium chloride). Covalent bonding involves the sharing of electrons between atoms. This typically occurs between nonmetals. Example: H₂O (water).
Question 7: Predicting Ionic Charges
Question: Predict the charge of the ion formed by each of the following elements: (a) Potassium (K), (b) Sulfur (S), (c) Aluminum (Al).
Answer:
(a) Potassium (K) is an alkali metal and typically loses one electron to form a +1 ion (K⁺). (b) Sulfur (S) is a nonmetal and typically gains two electrons to form a -2 ion (S²⁻). (c) Aluminum (Al) is a metal and typically loses three electrons to form a +3 ion (Al³⁺).
Advanced Concepts and Applications
This section delves into more complex topics related to atoms and ions, further solidifying your understanding.
Polyatomic Ions
Polyatomic ions are groups of atoms that carry a net electrical charge. They behave as single units in chemical reactions and form ionic bonds with other ions. Examples include sulfate (SO₄²⁻), nitrate (NO₃⁻), and ammonium (NH₄⁺).
Oxidation States and Redox Reactions
Oxidation states (or oxidation numbers) represent the hypothetical charge an atom would have if all bonds were completely ionic. They are useful in balancing redox (reduction-oxidation) reactions, where electrons are transferred between atoms. Oxidation involves the loss of electrons, while reduction involves the gain of electrons.
Electrolytes and Conductivity
Electrolytes are substances that, when dissolved in water, produce ions that can conduct electricity. Ionic compounds are typically good electrolytes because they dissociate into ions in solution. The conductivity of a solution depends on the concentration of ions present.
Applications of Atoms and Ions
Understanding atoms and ions is crucial in many fields, including:
- Medicine: Many medications involve ionic compounds or are administered as ions.
- Materials Science: The properties of materials are closely related to their atomic and ionic structure.
- Environmental Science: Ionic processes are essential in understanding pollution and environmental remediation.
- Agriculture: Nutrient uptake by plants involves ionic processes.
Conclusion
This comprehensive guide provides a thorough understanding of atoms and ions, covering fundamental concepts and more advanced topics. By understanding atomic structure, ionic bonding, and the properties of ions, you can confidently tackle any atoms and ions worksheet and build a solid foundation for further studies in chemistry. Remember to practice regularly and seek clarification on any areas you find challenging. This dedicated effort will ensure mastery of these fundamental concepts.
Latest Posts
Latest Posts
-
Foundations In Health And Safety E Learning Post Test
Apr 04, 2025
-
Circuit Training Chain Rule Worksheet Answers
Apr 04, 2025
-
Hesi Case Study Management Of A Surgical Unit
Apr 04, 2025
-
Which Size Memory Modules Were Installed
Apr 04, 2025
-
Una Carta Para Mama Worksheet Answers
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Atoms And Ions Worksheet Answer Key . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
