Lab 1 Measurement Pipette Use And The Standard Curve
Onlines
Apr 02, 2025 · 8 min read
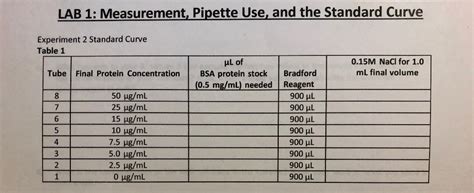
Table of Contents
Lab 1: Measurement Pipette Use and the Standard Curve
This comprehensive guide delves into the crucial laboratory techniques of using measurement pipettes and constructing standard curves. These are foundational skills in analytical chemistry and biochemistry, essential for accurate and reliable experimental results. We'll explore the practical aspects of pipette operation, potential sources of error, and the importance of meticulous technique. Furthermore, we'll dissect the creation and interpretation of standard curves, illustrating their vital role in quantitative analysis.
Understanding Measurement Pipettes
Measurement pipettes, unlike volumetric pipettes, allow for the dispensing of variable volumes of liquid. They're graduated along their length, enabling the precise measurement and delivery of a range of volumes within their capacity. Commonly available in glass or plastic, these pipettes are essential tools in any laboratory setting.
Types of Measurement Pipettes
Several types of measurement pipettes exist, each with its own design and application:
-
Mohr Pipettes: These pipettes have graduations that extend to the tip, allowing for the dispensing of the entire contained volume. However, the last drop should not be blown out as it's part of the calibrated volume.
-
Serological Pipettes: These pipettes have graduations that extend beyond the tip, permitting the delivery of the full volume, including the remaining liquid in the tip. The final drop is usually blown out using a pipette bulb or a pipette filler.
-
Pasteur Pipettes: These are simple, disposable pipettes typically made of plastic and used for transferring small volumes of liquids. They lack precise graduations and are mainly used for qualitative transfers.
Proper Pipette Usage
Accurate pipette use is paramount to obtaining reliable results. Follow these steps for optimal performance:
-
Choose the Correct Pipette: Select a pipette with a capacity appropriate for the desired volume. Using a pipette outside its range can lead to significant errors.
-
Pre-Rinse: Before drawing up the solution, rinse the pipette with a small amount of the solution to be measured. This ensures that the solution's concentration remains consistent and prevents dilution effects.
-
Filling the Pipette: Using a pipette bulb or a pipette filler (recommended for safety and accuracy), carefully draw the liquid into the pipette to the desired volume. Avoid introducing air bubbles, as they will affect the accuracy of the measurement.
-
Dispensing the Liquid: Slowly dispense the liquid into the receiving vessel. For Mohr pipettes, allow the liquid to drain under the influence of gravity. For serological pipettes, drain the liquid completely and carefully touch the tip against the inside of the vessel to expel the remaining liquid.
-
Calibration and Accuracy: It’s critical to understand the pipette’s calibration and tolerances. The markings on the pipette indicate the volume with associated uncertainty. Always consult the pipette's specifications.
Common Sources of Error in Pipetting
Several factors can introduce errors into pipette measurements:
-
Parallax Error: This occurs when the meniscus is not read at eye level. Ensure your eye is level with the meniscus to avoid this error.
-
Incorrect Pipette Technique: Improper filling, dispensing, or blowing out techniques can lead to significant errors. Strict adherence to the procedure is crucial.
-
Temperature Effects: Temperature changes can affect the viscosity of the liquid, potentially leading to inaccuracies. Maintain consistent temperature throughout the experiment.
-
Pipette Calibration: Over time, pipettes can become miscalibrated. Regular calibration checks are necessary to maintain accuracy.
-
Contamination: Cross-contamination between solutions can also compromise results. Always use clean and dry pipettes for each solution.
Understanding Standard Curves
A standard curve, also known as a calibration curve, is a graphical representation of the relationship between the known concentrations of a substance and their corresponding analytical signals. This curve is crucial for determining the concentration of an unknown sample based on its measured signal.
Constructing a Standard Curve
Creating a reliable standard curve involves several steps:
-
Prepare Standard Solutions: Prepare a series of solutions with known concentrations of the analyte (the substance being measured). This series is called a standard series or calibration series. The concentrations should span a range that encompasses the expected concentration of the unknown sample.
-
Measure the Analytical Signal: Using an appropriate analytical technique (e.g., spectrophotometry, chromatography), measure the signal produced by each standard solution. This signal is directly proportional to the concentration of the analyte.
-
Plot the Data: Plot the measured signals (y-axis) against the corresponding concentrations (x-axis). The resulting graph should show a linear relationship, although nonlinear standard curves can also be used if appropriate.
-
Determine the Equation of the Line: Using linear regression, determine the equation of the line (y = mx + c) that best fits the data points. This equation defines the standard curve and allows for the determination of unknown concentrations.
-
Determine the R² Value: The R² value, also known as the coefficient of determination, indicates the goodness of fit of the line to the data points. A higher R² value (closer to 1) indicates a better fit and therefore a more reliable standard curve.
Choosing the Appropriate Analytical Technique
The choice of analytical technique depends on the properties of the analyte and the desired sensitivity and accuracy. Common techniques used for generating standard curves include:
-
Spectrophotometry: This technique measures the absorbance or transmission of light through a solution, which is directly proportional to the concentration of the analyte. It is widely used for determining the concentration of colored solutions or solutions that can be made colored using appropriate reagents.
-
Chromatography: This technique separates the components of a mixture based on their different physical or chemical properties. The concentration of the analyte can then be determined by measuring the area under its peak in the chromatogram. Various chromatographic techniques like HPLC and GC are used depending on the nature of the analyte.
-
Electrochemical Methods: Techniques like potentiometry or voltammetry measure the electrical properties of the solution and relate it to the analyte concentration. These methods are often used for ionic species.
Using the Standard Curve to Determine Unknown Concentrations
Once a reliable standard curve has been generated, it can be used to determine the concentration of an unknown sample. Simply measure the analytical signal of the unknown sample using the same technique used to generate the standard curve. Then, using the equation of the line from the standard curve, substitute the measured signal (y) and solve for the concentration (x).
Interpreting Standard Curves and Assessing Quality
A well-constructed standard curve will exhibit a strong linear relationship between the concentration and the analytical signal. Several factors indicate the quality of a standard curve:
-
Linearity: The data points should fall along a straight line over the concentration range used. Non-linearity may indicate interference, limitations of the technique or concentration outside the linear range.
-
R² value: A high R² value (close to 1) indicates a good fit of the data to the linear model.
-
Outliers: Any data points that deviate significantly from the linear trend should be investigated. Potential causes could include errors in measurement or sample preparation.
-
Range: The standard curve should cover a concentration range that includes the expected concentration of the unknown samples. Extrapolation beyond the range of the standard curve should be avoided, as it is less reliable.
Potential Sources of Error in Standard Curves
Several factors can affect the accuracy and reliability of standard curves:
-
Sample Preparation Errors: Incorrect preparation of standard solutions or unknown samples can lead to significant errors. Meticulous attention to detail is crucial.
-
Instrumental Errors: Malfunctioning instruments or incorrect instrument settings can affect the accuracy of the measurements. Regular instrument calibration and maintenance are important.
-
Interferences: Other substances in the sample or standard solutions may interfere with the measurement of the analyte, leading to inaccurate results. Appropriate sample preparation techniques, like filtration or extraction, may mitigate this issue.
Combining Pipette Use and Standard Curves in Experiments
The effective use of measurement pipettes is intimately tied to the creation and application of standard curves. In many experiments, accurate pipetting is crucial for creating the standard solutions needed to generate the standard curve. Any errors introduced during pipetting will directly affect the accuracy of the standard curve and subsequently, the concentration determination of the unknown sample.
For instance, in a spectrophotometric analysis to quantify protein concentration using the Bradford assay, precise pipetting of protein standards and samples is critical for generating a reliable standard curve. Inaccurate pipetting will lead to inaccurate absorbance readings and ultimately, an erroneous protein concentration determination.
Similarly, in enzyme kinetics studies, accurate pipetting of enzyme solutions and substrates is necessary for creating the standard curve to determine the enzyme activity. Small errors in pipetting can cause significant deviations in the observed rates, compromising the accuracy of the derived kinetic parameters.
Conclusion
Mastering the techniques of using measurement pipettes and constructing standard curves is essential for any aspiring scientist. These fundamental skills are critical for accurately measuring solutions and performing quantitative analysis. Careful attention to detail, meticulous technique, and a thorough understanding of potential error sources are vital for obtaining reliable and reproducible experimental results. By consistently applying these methods and understanding their limitations, researchers can significantly improve the accuracy and reliability of their experimental data and strengthen the validity of their conclusions. The information provided in this guide provides a foundation for building proficiency in these important laboratory techniques. Remember that practice and a focus on accuracy are key to becoming proficient in these essential laboratory skills.
Latest Posts
Latest Posts
-
Aba Teamwork Makes The App Work
Apr 03, 2025
-
Chapter 5 Summary Of Into The Wild
Apr 03, 2025
-
Chapter 10 Catcher In The Rye Summary
Apr 03, 2025
-
Fundamentals Of Electric Circuits 7th Edition Solutions Pdf Free
Apr 03, 2025
-
Free Particle Model Worksheet 2 Interactions Answer Key
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Lab 1 Measurement Pipette Use And The Standard Curve . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
