Match Each Description With The Appropriate Step In Enzyme Catalysis.
Onlines
Apr 02, 2025 · 6 min read
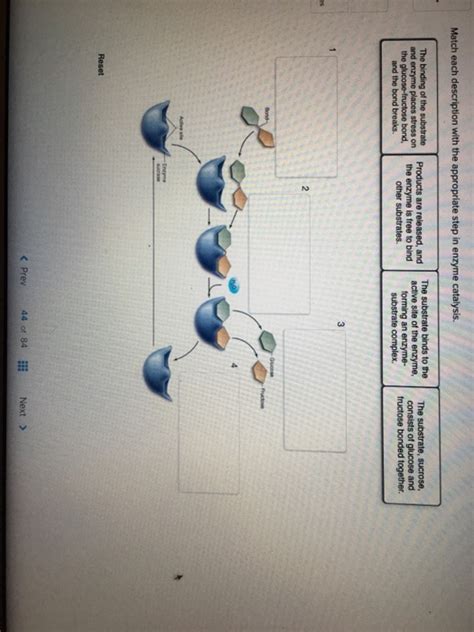
Table of Contents
Match Each Description with the Appropriate Step in Enzyme Catalysis
Enzymes are biological catalysts that dramatically accelerate the rate of virtually all chemical reactions within cells. Understanding the precise steps involved in enzyme catalysis is crucial to comprehending cellular processes and developing new therapies. This article will delve into the detailed mechanisms of enzyme catalysis, matching specific descriptions with the corresponding steps in the process. We'll explore the key players involved – enzymes, substrates, and the enzyme-substrate complex – and how they interact to achieve remarkable catalytic efficiency.
The Stages of Enzyme Catalysis: A Detailed Breakdown
Enzyme catalysis is a multi-step process, typically involving these key stages:
- Substrate Binding: The enzyme and substrate come together to form an enzyme-substrate complex.
- Transition State Formation: The substrate undergoes structural changes to reach a high-energy transition state.
- Product Formation: The transition state collapses, forming the product(s).
- Product Release: The product(s) detach from the enzyme, leaving the enzyme free to catalyze another reaction.
Let's examine each stage in more detail, linking specific descriptions to their appropriate step.
1. Substrate Binding: The Initial Encounter
This initial step is critical for enzyme function. The enzyme possesses a unique three-dimensional structure, including a specific region called the active site. This active site is where the substrate binds, initiating the catalytic process. The active site's shape and chemical properties are highly complementary to the substrate, ensuring a highly specific interaction. This specificity prevents the enzyme from reacting with inappropriate molecules, maintaining cellular order and efficiency.
Descriptions matching Substrate Binding:
- "The enzyme's active site interacts with the substrate, forming a temporary complex." This perfectly describes the formation of the enzyme-substrate complex. The temporary nature emphasizes the transient interaction necessary for catalysis.
- "The substrate binds to a specific region on the enzyme, typically a cleft or pocket." This highlights the active site's location and its role as a binding pocket for the substrate.
- "Weak non-covalent interactions (e.g., hydrogen bonds, van der Waals forces) stabilize the enzyme-substrate complex." These interactions ensure the substrate remains bound long enough for the reaction to proceed, without being too tightly bound to inhibit release of the product.
- "Conformational changes in the enzyme occur upon substrate binding, inducing a better fit (induced fit model)." This describes the dynamic nature of the enzyme-substrate interaction, emphasizing the enzyme's flexibility and adaptability. The induced fit model refines the lock-and-key model, which suggests a rigid, pre-formed active site.
2. Transition State Formation: The Energy Barrier
The transition state is a high-energy, unstable intermediate state between the substrate and the product. Reaching this transition state is the rate-limiting step in the reaction. Enzymes significantly lower the activation energy required to reach this transition state, thus dramatically increasing the reaction rate. They achieve this by stabilizing the transition state, making it energetically more accessible.
Descriptions matching Transition State Formation:
- "The substrate undergoes a conformational change, reaching a high-energy intermediate." This describes the pivotal role of the transition state in the reaction pathway.
- "The enzyme stabilizes the transition state, reducing the activation energy." This explains the enzyme's catalytic mechanism, highlighting its role in lowering the energy barrier.
- "Bond breaking and bond formation occur within the enzyme-substrate complex, leading to the transition state." This focuses on the chemical transformations happening within the active site, ultimately forming the transition state.
- "Specific amino acid residues in the active site interact with the substrate, facilitating the transition state formation." This emphasizes the importance of the active site's chemical environment in stabilizing the transition state.
- "The enzyme provides a microenvironment that optimizes the conditions for transition state formation." This highlights the active site's function in creating ideal conditions, such as optimal pH or polarity, for the reaction to proceed.
3. Product Formation: The Transformation
Once the transition state is reached, the reaction proceeds relatively quickly to product formation. The bonds in the substrate are rearranged, resulting in the creation of new bonds and the formation of the product(s). The enzyme facilitates this transformation by precisely orienting the substrate molecules and providing the necessary catalytic groups for the reaction.
Descriptions matching Product Formation:
- "The high-energy transition state collapses, leading to the formation of product(s)." This succinctly describes the outcome of reaching the transition state.
- "New chemical bonds are formed, and existing bonds are broken, resulting in the formation of the product(s)." This focuses on the chemical changes that occur during product formation.
- "The enzyme facilitates bond rearrangement by precisely positioning the substrate molecules." This highlights the enzyme's role in optimizing the reaction's efficiency.
- "The enzyme's active site provides catalytic groups that assist in bond breaking and bond formation." This emphasizes the catalytic contribution of specific amino acid residues within the active site.
4. Product Release: The Final Step
After the product(s) are formed, they must dissociate from the enzyme to allow the enzyme to catalyze another reaction. The binding affinity of the enzyme for the product is typically lower than for the substrate, facilitating this release. The release of the product regenerates the free enzyme, completing the catalytic cycle.
Descriptions matching Product Release:
- "The product(s) dissociate from the enzyme, leaving the enzyme free to catalyze another reaction." This is the direct description of the product release step.
- "The enzyme returns to its original conformation after product release." This highlights the cyclical nature of enzyme catalysis.
- "Weak non-covalent interactions between the enzyme and product are broken, leading to product dissociation." This emphasizes the nature of the interactions between the enzyme and the released product.
- "The conformational change that occurred upon substrate binding is reversed upon product release." This connects the conformational changes throughout the catalytic cycle, highlighting its dynamic nature.
Enzyme Kinetics and Catalytic Efficiency
The efficiency of enzyme catalysis is often described using kinetic parameters, such as K<sub>m</sub> (Michaelis constant) and k<sub>cat</sub> (turnover number). K<sub>m</sub> reflects the enzyme's affinity for its substrate, while k<sub>cat</sub> represents the number of substrate molecules converted to product per enzyme molecule per unit time. High k<sub>cat</sub>/K<sub>m</sub> ratios indicate high catalytic efficiency. Understanding these parameters is crucial for assessing enzyme activity and designing effective enzyme-based therapies.
Factors Affecting Enzyme Activity
Several factors can influence the rate of enzyme-catalyzed reactions, including:
- Temperature: Enzymes generally have an optimal temperature for activity. High temperatures can denature the enzyme, while low temperatures can slow down the reaction rate.
- pH: Enzymes have an optimal pH range, deviations from which can alter the enzyme's three-dimensional structure and reduce its activity.
- Substrate concentration: Increasing substrate concentration typically increases reaction rate up to a point where the enzyme becomes saturated.
- Enzyme concentration: Increasing enzyme concentration increases the rate of reaction.
- Inhibitors: Molecules that interfere with enzyme activity, either competitively or non-competitively.
- Activators: Molecules that enhance enzyme activity, often by binding to allosteric sites and inducing conformational changes.
Conclusion: The Intricate Dance of Enzyme Catalysis
Enzyme catalysis is a sophisticated and highly regulated process. By understanding the specific steps involved, from substrate binding to product release, and the factors influencing enzyme activity, we can gain deeper insights into the intricate mechanisms driving life's biochemical reactions. This knowledge forms the basis for various applications, including the development of new drugs, diagnostic tools, and industrial biocatalysts. The remarkable efficiency and specificity of enzymes continue to inspire research and innovation in the field of biochemistry and beyond. Further research into the finer details of each step and the interactions involved will continue to refine our understanding of this fundamental biological process.
Latest Posts
Latest Posts
-
How Many Chapters Are In Crime And Punishment
Apr 03, 2025
-
How To Know My Glo Number
Apr 03, 2025
-
The Blank Causes The Stage To Move Upward Or Downward
Apr 03, 2025
-
Which Statement Shows That Money Is A Measure Of Value
Apr 03, 2025
-
Pot Syel La Va Louvri Pou Mwen Lyrics
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Match Each Description With The Appropriate Step In Enzyme Catalysis. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
