The Products Of A Combustion Reaction Do Not Include ____.
Onlines
Apr 02, 2025 · 5 min read
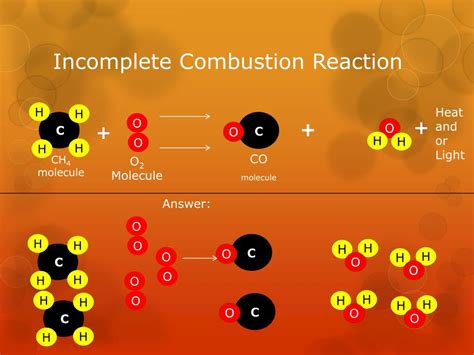
Table of Contents
The Products of a Combustion Reaction Do Not Include Unburnt Reactants
Combustion, a cornerstone of many industrial processes and a fundamental aspect of everyday life, is a rapid chemical reaction between a substance and an oxidant, usually oxygen, that produces heat and light. Understanding the products of this reaction is crucial in various fields, from engineering and environmental science to medicine and culinary arts. This comprehensive article explores the typical products of combustion, highlighting what is conspicuously absent from a complete combustion process: unburnt reactants.
Understanding Combustion: A Closer Look
Before diving into what's missing from a complete combustion reaction, let's establish a firm grasp of the process itself. Combustion is fundamentally an exothermic redox reaction, meaning it releases energy in the form of heat and involves the transfer of electrons. The reaction typically involves a fuel (the substance being oxidized) and an oxidant (usually oxygen), often accompanied by an ignition source to initiate the reaction.
Types of Combustion
The completeness of combustion significantly affects the products formed. We can categorize combustion into two main types:
-
Complete Combustion: This occurs when sufficient oxygen is present to fully oxidize the fuel. The result is a relatively clean and predictable set of products.
-
Incomplete Combustion: This happens when the oxygen supply is insufficient. This leads to the formation of various partially oxidized products, resulting in a less efficient energy release and potentially harmful byproducts.
Typical Products of Complete Combustion
In a complete combustion reaction involving hydrocarbons (fuels containing only carbon and hydrogen), the primary products are carbon dioxide (CO₂) and water (H₂O). The reaction can be generalized as follows:
CxHy + (x + y/4)O₂ → xCO₂ + (y/2)H₂O + Heat
Where:
- CxHy represents the hydrocarbon fuel
- O₂ represents oxygen
- CO₂ represents carbon dioxide
- H₂O represents water
For example, the complete combustion of methane (CH₄) is:
CH₄ + 2O₂ → CO₂ + 2H₂O + Heat
In addition to CO₂ and H₂O, complete combustion might also produce:
- Heat: The primary energy output of combustion reactions.
- Light: Visible light is produced during combustion, especially in rapid or intense reactions like flames.
- Minor amounts of other gases: Depending on the fuel and conditions, trace amounts of other gases may form, but these are usually negligible in a well-controlled combustion process.
What the Products Don't Include: The Absence of Unburnt Reactants
The key takeaway is that a complete combustion reaction efficiently consumes all the reactants. Therefore, unburnt reactants, whether the fuel or the oxidant (oxygen), are notably absent from the products of a complete combustion process. This is the defining characteristic that differentiates complete from incomplete combustion.
The Significance of Complete Combustion
The absence of unburnt reactants is directly linked to the efficiency and environmental impact of combustion. Complete combustion signifies maximized energy extraction from the fuel. It also minimizes the formation of pollutants. Unburnt fuel contributes to air pollution, and incomplete combustion creates various harmful byproducts.
The Implications of Incomplete Combustion
Incomplete combustion, as mentioned earlier, occurs when there's insufficient oxygen to fully oxidize the fuel. This results in the presence of unburnt reactants in the combustion products alongside several other undesirable compounds:
- Carbon Monoxide (CO): A highly toxic gas that is a significant air pollutant.
- Soot (Elemental Carbon): Fine particles of carbon that contribute to air pollution and respiratory problems.
- Hydrocarbons (Unburnt Fuel): These unburnt fuel molecules contribute to air pollution and greenhouse gas emissions.
- Other Partially Oxidized Compounds: A variety of other partially oxidized compounds can form, depending on the nature of the fuel and the combustion conditions.
Factors Affecting Combustion Completeness
Several factors influence the completeness of a combustion reaction:
-
Oxygen Supply: Sufficient oxygen is paramount for complete combustion. An oxygen-deficient environment inevitably leads to incomplete combustion.
-
Fuel-to-Air Ratio: The proper ratio of fuel to air is crucial. An overly rich fuel mixture (too much fuel relative to oxygen) results in incomplete combustion, while a lean mixture (too much air) may not be as efficient.
-
Mixing: Thorough mixing of fuel and air is vital to ensure uniform combustion and prevent localized areas of incomplete combustion.
-
Temperature: Sufficient temperature is essential to initiate and maintain the combustion reaction. Low temperatures can lead to incomplete burning.
-
Pressure: Pressure can influence the reaction rate and the mixing of reactants, affecting combustion efficiency.
Real-World Examples and Applications
The concept of complete combustion and the absence of unburnt reactants is crucial across various applications:
-
Internal Combustion Engines: Modern car engines are designed to achieve near-complete combustion, maximizing fuel efficiency and minimizing harmful emissions. Engine management systems carefully control the fuel-to-air ratio and other parameters to optimize combustion.
-
Power Plants: Large-scale power plants that burn fossil fuels aim for complete combustion to produce maximum energy with minimal pollution. Sophisticated emission control technologies are employed to further reduce pollutants.
-
Industrial Furnaces and Kilns: The control of combustion parameters in industrial processes is critical for both efficiency and product quality. Complete combustion ensures consistent temperatures and minimizes emissions.
-
Cooking: While not as strictly controlled as industrial processes, efficient cooking depends on achieving complete combustion in the flame, ensuring even heat distribution and avoiding harmful byproducts.
Conclusion: The Importance of Complete Combustion
The absence of unburnt reactants is a defining characteristic of complete combustion. Achieving complete combustion is crucial for several reasons:
-
Maximized Energy Efficiency: Complete combustion extracts the maximum amount of energy from the fuel.
-
Minimized Pollution: It reduces or eliminates the formation of harmful pollutants such as CO, soot, and unburnt hydrocarbons.
-
Improved Process Control: Understanding and managing the factors that influence combustion completeness are critical for efficient and safe operation in various industrial and domestic applications.
In essence, the products of a combustion reaction should ideally consist only of the oxidized products of the fuel and oxidant, primarily carbon dioxide and water in the case of hydrocarbon fuels. The absence of unburnt reactants signifies optimal combustion efficiency and environmental responsibility. Any presence of unburnt fuel highlights the need for improved combustion control to enhance efficiency and mitigate harmful emissions.
Latest Posts
Latest Posts
-
The Paper Is Stating The Poems Summaries Themes Topics Transitions
Apr 03, 2025
-
Hw 7 1 1 3 Arithmetic And Geometric Sequences
Apr 03, 2025
-
A Client With Copd Has A Blood Ph Of 7 25
Apr 03, 2025
-
What Is The Best Way To Prevent Ratio Strain
Apr 03, 2025
-
Answer The Following Questions In Brief
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about The Products Of A Combustion Reaction Do Not Include ____. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
