Ap Chem Unit 4 Progress Check Frq
Onlines
Apr 04, 2025 · 5 min read
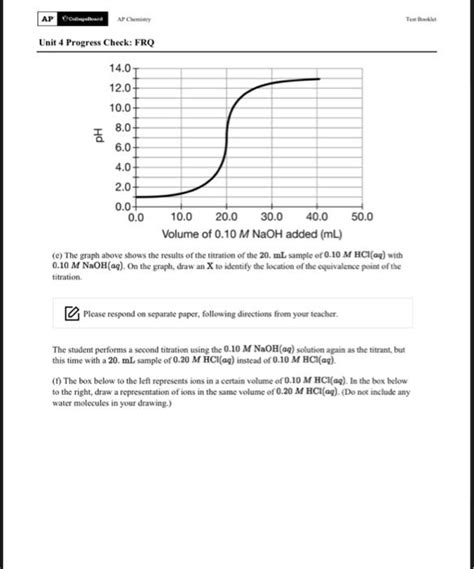
Table of Contents
AP Chem Unit 4 Progress Check: FRQ Mastery Guide
Unit 4 of AP Chemistry, encompassing the intricacies of reactions, is notoriously challenging. The free-response questions (FRQs) on the AP Chemistry exam within this unit often prove to be a significant hurdle for students. This comprehensive guide will dissect the common themes, provide effective problem-solving strategies, and equip you with the knowledge to confidently tackle these challenging questions. We'll explore various question types, offer sample problems with detailed solutions, and provide tips for maximizing your score.
Understanding the Unit 4 Focus: Reactions
Unit 4 centers around the fundamental principles governing chemical reactions. Key concepts include:
-
Types of Reactions: You must be comfortable identifying and classifying reactions as acid-base, redox (reduction-oxidation), precipitation, and complex ion formation. Understanding the driving forces behind these reactions (e.g., enthalpy changes, entropy changes, equilibrium considerations) is crucial.
-
Stoichiometry: Mastering stoichiometric calculations is essential. This includes balancing chemical equations, converting between moles and grams, determining limiting reactants, and calculating theoretical and percent yields. Expect questions involving both simple and complex reaction schemes.
-
Thermochemistry: A deep understanding of enthalpy (ΔH), entropy (ΔS), and Gibbs Free Energy (ΔG) is paramount. You'll need to apply these concepts to predict reaction spontaneity and calculate equilibrium constants. Hess's Law and standard enthalpy of formation calculations are frequently tested.
-
Equilibrium: Understanding equilibrium constants (K), Le Chatelier's Principle, and the relationship between K and ΔG is crucial. Be prepared to analyze how changes in concentration, temperature, and pressure affect equilibrium positions.
-
Kinetics: While not the primary focus, understanding basic concepts like reaction rates and rate laws can be incorporated into Unit 4 FRQs, especially those involving equilibrium considerations.
-
Acid-Base Equilibria: This is a significant part of Unit 4. You must be proficient in calculating pH, pOH, and concentrations of various species in buffer solutions and weak acid/base systems. Understanding titration curves and their interpretation is vital.
Common FRQ Question Types in Unit 4
Unit 4 FRQs typically incorporate multiple concepts within a single question, requiring a multi-step approach. Here are common question types:
1. Reaction Identification and Stoichiometry
These questions present a reaction scenario and ask you to:
- Identify the type of reaction. Is it a redox reaction, acid-base neutralization, precipitation, or complex ion formation?
- Balance the chemical equation. Accuracy in balancing is critical as it impacts subsequent calculations.
- Perform stoichiometric calculations. Determine limiting reactants, theoretical yield, percent yield, etc.
Example: A reaction between 20.0 g of silver nitrate and 15.0 g of sodium chloride produces silver chloride precipitate and sodium nitrate. Identify the type of reaction, balance the equation, determine the limiting reactant, and calculate the theoretical yield of silver chloride.
2. Thermochemistry and Equilibrium Calculations
These questions often involve:
- Calculating enthalpy changes. Use Hess's Law, standard enthalpies of formation, or calorimetry data.
- Predicting reaction spontaneity. Using ΔG = ΔH - TΔS, determine whether a reaction is spontaneous under given conditions.
- Equilibrium calculations. Determine equilibrium concentrations, K values, or the effect of changes on the equilibrium position using ICE (Initial, Change, Equilibrium) tables.
Example: Given the standard enthalpies of formation for reactants and products, calculate the ΔH for a given reaction. Determine if the reaction is spontaneous at 298 K, given the ΔS. Explain your reasoning using Le Chatelier's Principle.
3. Acid-Base Equilibria Problems
These questions assess your understanding of acid-base chemistry:
- pH and pOH calculations. Determine pH for strong and weak acids and bases, using appropriate equilibrium expressions.
- Buffer solutions. Calculate the pH of a buffer solution and analyze its response to the addition of strong acids or bases.
- Titration curves. Interpret titration curves, identifying equivalence points and buffering regions.
Example: A 25.0 mL solution of 0.100 M acetic acid is titrated with 0.100 M NaOH. Calculate the pH at the following points: before the addition of any NaOH, at half the equivalence point, and at the equivalence point. Sketch the titration curve.
4. Integrated Problems
These are the most challenging FRQs, combining multiple concepts from throughout Unit 4. They often involve complex scenarios that require a systematic approach and strong problem-solving skills. These problems assess your ability to apply knowledge in novel situations.
Example: A reaction involving the formation of a complex ion is carried out at various temperatures. Experimental data is provided, including equilibrium constants at different temperatures. The questions might ask you to:
- Determine the type of reaction.
- Calculate ΔH and ΔS for the reaction using the van't Hoff equation (lnK = -ΔH/R(1/T) + ΔS/R).
- Analyze the spontaneity of the reaction at different temperatures.
- Predict the effect of changes in concentration or temperature on the equilibrium position.
Strategies for Mastering Unit 4 FRQs
- Master the fundamental concepts: Ensure a thorough understanding of all the topics listed above. Don't just memorize formulas; understand their underlying principles.
- Practice, practice, practice: Work through numerous practice problems, including those from past AP Chemistry exams. This is the most crucial aspect of mastering these questions.
- Develop a systematic approach: When tackling FRQs, use a step-by-step approach, clearly showing your work and explaining your reasoning. Label diagrams and tables.
- Learn to interpret data: Many FRQs present data in tables or graphs. Practice analyzing this data to extract relevant information.
- Seek feedback: If possible, have a teacher or tutor review your work to identify areas for improvement.
- Understand the scoring rubric: Familiarize yourself with the AP Chemistry scoring rubric to understand how points are awarded for each part of the FRQ. This will help you prioritize your efforts and avoid losing easy points due to incomplete answers or missing units.
- Manage your time effectively: During the AP exam, allocate sufficient time to each FRQ. Practice working under time constraints to improve efficiency.
- Use appropriate units and significant figures: Pay close attention to units and maintain the appropriate number of significant figures throughout your calculations. Points are often deducted for errors in this area.
Conclusion
Successfully navigating the AP Chemistry Unit 4 Progress Check FRQs requires a deep understanding of chemical reactions, meticulous calculation skills, and the ability to apply these concepts to complex scenarios. By mastering the concepts outlined in this guide, practicing consistently, and developing a structured problem-solving approach, you can significantly improve your performance on these challenging questions and achieve your desired score on the AP Chemistry exam. Remember, consistent effort and practice are key to success. Good luck!
Latest Posts
Latest Posts
-
Unlike Traditional Training Programs Action Learning
Apr 05, 2025
-
Evolution In Action Graphing And Statistics
Apr 05, 2025
-
Energy And Specific Heat Report Sheet
Apr 05, 2025
-
Persuasive Techniques In The Market Chapter 5 Lesson 3
Apr 05, 2025
-
Which Of The Following Segments Is A Diameter Of 0
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Ap Chem Unit 4 Progress Check Frq . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
