Match Each Description With The Correct Level Of Protein Structure.
Onlines
Apr 02, 2025 · 6 min read
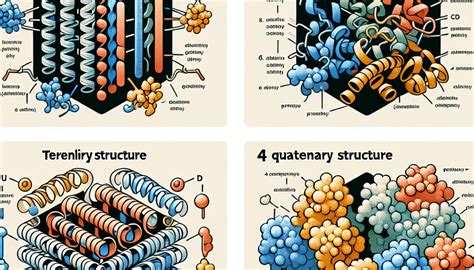
Table of Contents
Match Each Description with the Correct Level of Protein Structure
Proteins are the workhorses of the cell, carrying out a vast array of functions crucial for life. Understanding their structure is key to understanding their function. Protein structure is a hierarchical system, with four main levels: primary, secondary, tertiary, and quaternary. Each level builds upon the previous one, creating a complex and intricate three-dimensional architecture. This article will delve into each level of protein structure, providing detailed descriptions and matching them to specific characteristics.
Primary Structure: The Linear Sequence of Amino Acids
The primary structure of a protein is its simplest and most fundamental level. It's essentially the linear sequence of amino acids linked together by peptide bonds. Think of it as the alphabet of protein structure; the specific order of these amino acids determines all subsequent levels of structure and, ultimately, the protein's function. This sequence is dictated by the genetic code, specifically the sequence of nucleotides in the gene encoding that particular protein. Any change in this sequence, such as a single amino acid substitution (a mutation), can have profound consequences, sometimes leading to protein malfunction or disease. For example, a single amino acid change in the hemoglobin protein causes sickle cell anemia.
Key Characteristics of Primary Structure:
- Peptide bonds: Covalent bonds linking the carboxyl group of one amino acid to the amino group of the next.
- Amino acid sequence: The specific order of amino acids, written from the N-terminus (amino group) to the C-terminus (carboxyl group).
- Genetic code: The DNA sequence determines the amino acid sequence.
- Unique to each protein: Every protein has a unique primary structure.
Example Description Matching Primary Structure: "The precise order of amino acids in a polypeptide chain, determined genetically."
Secondary Structure: Local Folding Patterns
The secondary structure refers to the local folding patterns of the polypeptide chain stabilized by hydrogen bonds between the backbone atoms (not the side chains). These patterns arise from interactions between the carbonyl oxygen (C=O) of one peptide bond and the amide hydrogen (N-H) of another peptide bond within the same polypeptide chain. Two common secondary structures are:
1. Alpha-Helices: These are coiled structures resembling a spiral staircase. The hydrogen bonds form between the carbonyl oxygen of one amino acid and the amide hydrogen of the amino acid four residues down the chain. This creates a stable, rod-like structure.
2. Beta-Sheets: These are formed by several segments of the polypeptide chain (beta-strands) arranged side-by-side, forming a pleated sheet-like structure. Hydrogen bonds form between carbonyl oxygens and amide hydrogens on adjacent beta-strands. Beta-sheets can be parallel (beta-strands run in the same direction) or antiparallel (beta-strands run in opposite directions).
Key Characteristics of Secondary Structure:
- Hydrogen bonds: Stabilizing forces between backbone atoms.
- Local folding patterns: Relatively short segments of the polypeptide chain.
- Alpha-helices and beta-sheets: Common structural motifs.
- Independent of side chains: Primarily determined by interactions between the polypeptide backbone.
Example Description Matching Secondary Structure: "Regular, repeating patterns of polypeptide chain folding, such as alpha-helices and beta-sheets, stabilized by hydrogen bonds."
Tertiary Structure: The Three-Dimensional Arrangement
The tertiary structure represents the three-dimensional arrangement of the entire polypeptide chain, including all its secondary structure elements. It is the overall folding of the protein, bringing distant parts of the polypeptide chain into close proximity. This folding is driven by a variety of forces, including:
- Hydrophobic interactions: Nonpolar amino acid side chains cluster together in the protein's interior, away from the surrounding water.
- Hydrogen bonds: Occur between side chains of various amino acids.
- Ionic bonds (salt bridges): Electrostatic interactions between oppositely charged amino acid side chains.
- Disulfide bonds: Covalent bonds between cysteine residues, creating strong cross-links within the protein.
These interactions create a unique three-dimensional shape that is essential for the protein's function. The tertiary structure is often described as the protein's "native state," representing its biologically active conformation.
Key Characteristics of Tertiary Structure:
- Three-dimensional folding: The complete spatial arrangement of the polypeptide chain.
- Multiple types of interactions: Hydrophobic interactions, hydrogen bonds, ionic bonds, and disulfide bonds.
- Globular or fibrous proteins: Proteins can adopt various shapes based on their tertiary structure.
- Functional conformation: The tertiary structure is essential for the protein's biological activity.
Example Description Matching Tertiary Structure: "The overall three-dimensional structure of a single polypeptide chain, including its folding and the spatial arrangement of its side chains."
Quaternary Structure: The Assembly of Subunits
The quaternary structure applies only to proteins composed of multiple polypeptide chains (subunits). It describes how these subunits interact and arrange themselves to form a functional protein complex. Each subunit may have its own primary, secondary, and tertiary structure, but the quaternary structure defines how these subunits associate with each other. The interactions between subunits are similar to those seen in tertiary structure – hydrophobic interactions, hydrogen bonds, ionic bonds, and disulfide bonds. Some proteins require multiple subunits to be functional; others may exist in different forms (isoforms) with varying numbers of subunits.
Key Characteristics of Quaternary Structure:
- Multiple polypeptide chains: Proteins with more than one subunit.
- Subunit interactions: Forces holding subunits together.
- Functional complexes: The quaternary structure is essential for the protein's activity.
- Symmetrical or asymmetrical: Subunits can arrange themselves in various patterns.
Example Description Matching Quaternary Structure: "The arrangement and interaction of multiple polypeptide chains in a multi-subunit protein complex."
Connecting Structure to Function: A Case Study of Enzymes
The relationship between protein structure and function is intimately linked. Consider enzymes, biological catalysts that speed up chemical reactions. Their unique three-dimensional shapes contain specific binding sites called active sites. The active site's precise arrangement of amino acid side chains allows it to bind to specific substrate molecules, the reactants in the enzyme-catalyzed reaction. Any disruption to the protein's structure, such as denaturation caused by heat or extreme pH, can alter the shape of the active site, preventing substrate binding and rendering the enzyme inactive.
For instance, a change in the primary structure (amino acid sequence) might lead to a change in the tertiary structure, altering the active site and thus affecting the enzyme's ability to catalyze the reaction. This highlights how the intricate hierarchy of protein structure ensures its biological function.
The Importance of Protein Folding and Misfolding
Proper protein folding is crucial for cellular function. Specialized molecular chaperones assist in the folding process, ensuring proteins achieve their correct three-dimensional structures. However, proteins can sometimes misfold, leading to the formation of aggregates that can disrupt cellular processes and contribute to various diseases, including Alzheimer's and Parkinson's disease. These misfolded proteins are often characterized by the exposure of hydrophobic regions that normally reside within the protein's core, leading to aggregation.
Summary Table: Matching Descriptions to Protein Structure Levels
| Protein Structure Level | Description |
|---|---|
| Primary | The linear sequence of amino acids in a polypeptide chain, determined by the genetic code. |
| Secondary | Local folding patterns of the polypeptide chain, such as alpha-helices and beta-sheets, stabilized by hydrogen bonds. |
| Tertiary | The overall three-dimensional structure of a single polypeptide chain, including its folding and side chain arrangement. |
| Quaternary | The arrangement and interaction of multiple polypeptide chains in a multi-subunit protein complex. |
This article provides a comprehensive overview of the four levels of protein structure. Understanding these levels is essential for comprehending the complex relationship between protein structure and function, as well as the implications of protein misfolding in disease. Further research into specific proteins and their structures can reveal even more fascinating details about the remarkable complexity of life's building blocks.
Latest Posts
Latest Posts
-
Nursing Care Plan For Neonatal Jaundice
Apr 03, 2025
-
Famous Forensic Christmas Mystery Picture Answer Key
Apr 03, 2025
-
Ecological Diversity Is A Measure Of The Number Of
Apr 03, 2025
-
The Mark Of True Sexual Deviation Is
Apr 03, 2025
-
Statistics For The Behavioral Sciences 10th Edition Pdf
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Match Each Description With The Correct Level Of Protein Structure. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
