For Each Pair Of Reactants Will There Be A Reaction
Onlines
Apr 01, 2025 · 6 min read
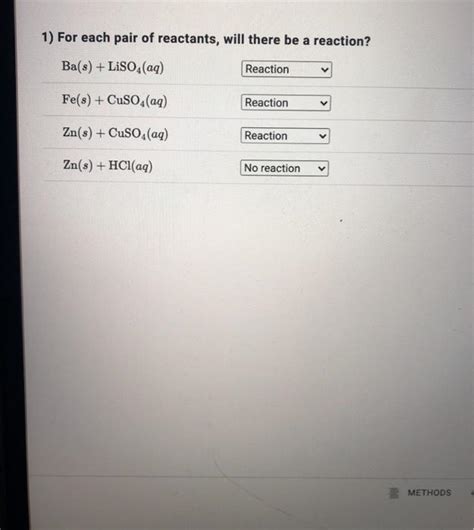
Table of Contents
Predicting Chemical Reactions: Will They React? A Comprehensive Guide
Predicting whether a chemical reaction will occur between two reactants is a fundamental concept in chemistry. It's not a simple yes or no answer, but rather a nuanced understanding based on several factors. This comprehensive guide delves into the various principles and considerations that dictate the likelihood of a reaction between any given pair of reactants. We'll explore the key factors, providing a robust framework for predicting reactivity.
Understanding Reactivity: The Driving Forces
The spontaneity of a chemical reaction hinges on several intertwined factors, primarily encompassing thermodynamics and kinetics.
Thermodynamics: The Energy Landscape
Thermodynamics helps us determine if a reaction can occur. The primary factor is the Gibbs Free Energy (ΔG). A negative ΔG indicates a spontaneous reaction, meaning it will proceed without external intervention. ΔG is determined by two other thermodynamic quantities:
- Enthalpy (ΔH): This represents the heat change during the reaction. Exothermic reactions (ΔH < 0) release heat, making them thermodynamically favorable.
- Entropy (ΔS): This reflects the change in disorder or randomness of the system. Reactions that increase entropy (ΔS > 0) are also thermodynamically favored.
The relationship between these is expressed as: ΔG = ΔH - TΔS, where T is the temperature in Kelvin. Even if ΔH is positive (endothermic), a large positive ΔS at high temperatures can still result in a negative ΔG, leading to a spontaneous reaction.
Kinetics: The Speed of Reaction
While thermodynamics tells us if a reaction can occur, kinetics dictates how fast it occurs. Even if a reaction is thermodynamically favorable (ΔG < 0), it might be impractically slow without sufficient activation energy.
- Activation Energy (Ea): This is the minimum energy required for the reactants to overcome the energy barrier and initiate the reaction. A high Ea leads to slow reaction rates, potentially making the reaction appear non-existent even if thermodynamically feasible.
- Reaction Rate: The speed at which reactants are converted into products. This is influenced by factors like concentration, temperature, pressure, and the presence of catalysts. Catalysts lower Ea, significantly accelerating reaction rates.
In summary: A reaction will only proceed if it's both thermodynamically feasible (ΔG < 0) and kinetically favorable (sufficiently fast).
Predicting Reactivity: A Multifaceted Approach
Predicting reactivity requires considering various factors, including:
1. The Nature of the Reactants:
- Electronegativity: The tendency of an atom to attract electrons in a chemical bond. Large differences in electronegativity can drive reactions, particularly redox reactions (electron transfer).
- Ionization Energy: The energy required to remove an electron from an atom. Elements with low ionization energies are more likely to lose electrons and participate in redox reactions.
- Electron Affinity: The energy change when an atom gains an electron. Elements with high electron affinities readily gain electrons.
- Oxidation States: The apparent charge of an atom in a compound. Changes in oxidation states indicate redox reactions.
2. Reaction Type: Identifying the Chemical Transformation
Recognizing the type of reaction provides valuable insight into its likelihood. Common reaction types include:
- Acid-Base Reactions: Involve the transfer of protons (H+ ions). Strong acids and bases react readily with each other. The strength of the acid and base determines the extent of the reaction.
- Precipitation Reactions: Occur when two soluble ionic compounds react to form an insoluble compound (precipitate). Solubility rules help predict the formation of precipitates.
- Redox Reactions: Involve the transfer of electrons. A strong oxidizing agent readily accepts electrons, while a strong reducing agent readily donates electrons.
- Combustion Reactions: Involve rapid reactions with oxygen, often producing heat and light. Combustible materials react readily with oxygen under appropriate conditions.
- Synthesis Reactions: Two or more substances combine to form a more complex substance.
- Decomposition Reactions: A single compound breaks down into two or more simpler substances.
- Single Displacement Reactions: One element replaces another in a compound.
- Double Displacement Reactions: Two compounds exchange ions to form two new compounds.
3. Reaction Conditions: Environmental Influences
The reaction environment plays a crucial role:
- Temperature: Higher temperatures usually increase reaction rates by providing more kinetic energy to overcome Ea.
- Pressure: Affects gaseous reactions; increased pressure can favor reactions that produce fewer gas molecules.
- Concentration: Higher concentrations of reactants lead to more frequent collisions and faster reaction rates.
- Solvent: The solvent can affect the solubility of reactants and the stability of intermediates, impacting the reaction outcome.
- Presence of Catalysts: Catalysts lower Ea without being consumed, accelerating reactions.
4. Using Activity Series and Solubility Rules: Practical Tools
Several tools simplify the prediction process:
- Activity Series: Lists metals in order of their reactivity. A more reactive metal can displace a less reactive metal from its compound.
- Solubility Rules: Predicts the solubility of ionic compounds in water. These rules help determine if a precipitate will form in a precipitation reaction.
Examples of Predicting Reactivity
Let's consider some examples to illustrate the concepts:
Example 1: Reaction of Sodium Metal (Na) with Water (H₂O)
- Nature of Reactants: Sodium is a highly reactive alkali metal with low ionization energy. Water is a polar molecule.
- Reaction Type: Redox reaction. Sodium loses an electron (oxidation), while water gains an electron (reduction).
- Thermodynamics: This reaction is highly exothermic (ΔH < 0) and increases entropy (ΔS > 0), resulting in a negative ΔG.
- Kinetics: The reaction is fast, even at room temperature.
- Prediction: A vigorous reaction occurs, producing sodium hydroxide (NaOH) and hydrogen gas (H₂).
Example 2: Reaction of Gold (Au) with Hydrochloric Acid (HCl)
- Nature of Reactants: Gold is a relatively unreactive metal. HCl is a moderately strong acid.
- Reaction Type: Potential redox reaction.
- Thermodynamics: The reaction is not thermodynamically favorable under standard conditions. Gold's high ionization energy makes it resistant to oxidation.
- Kinetics: Even if thermodynamically possible under certain conditions, the reaction would be extremely slow.
- Prediction: No significant reaction occurs under normal conditions.
Example 3: Reaction of Silver Nitrate (AgNO₃) and Sodium Chloride (NaCl)
- Nature of Reactants: Both are soluble ionic compounds.
- Reaction Type: Precipitation reaction.
- Thermodynamics: The formation of the insoluble silver chloride (AgCl) precipitate drives the reaction.
- Kinetics: The reaction is relatively fast.
- Prediction: A precipitate of silver chloride forms, leaving sodium nitrate (NaNO₃) in solution.
Advanced Techniques for Predicting Reactivity
For more complex reactions, advanced techniques are employed:
- Computational Chemistry: Uses computer simulations to model molecular interactions and predict reaction pathways and energetics.
- Quantum Mechanics: Provides a fundamental understanding of chemical bonding and reactivity at the atomic level.
Conclusion: A Holistic Approach
Predicting whether two reactants will undergo a chemical reaction requires a holistic approach, integrating thermodynamic considerations, kinetic factors, the nature of reactants, reaction type, and environmental conditions. While simple rules and guidelines exist, complex reactions often require advanced techniques to accurately predict their behavior. Understanding these principles empowers chemists to anticipate reaction outcomes, design efficient syntheses, and interpret experimental observations effectively. By considering the multiple facets discussed, you'll gain a much deeper understanding of the fascinating world of chemical reactivity.
Latest Posts
Latest Posts
-
Assisted Living Can Be Thought Of As A Combination Of
Apr 03, 2025
-
12 1 Identifying The Substance Of Genes
Apr 03, 2025
-
25 3 11 Packet Tracer Logging From Multiple Sources
Apr 03, 2025
-
Which Graph Represents Decreasing Distance With Increasing Time
Apr 03, 2025
-
The Lone Ranger And Tonto Fistfight In Heaven Analysis
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about For Each Pair Of Reactants Will There Be A Reaction . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
