Rank The Structures In Order Of Decreasing Electrophilic Strength
Onlines
Apr 04, 2025 · 5 min read
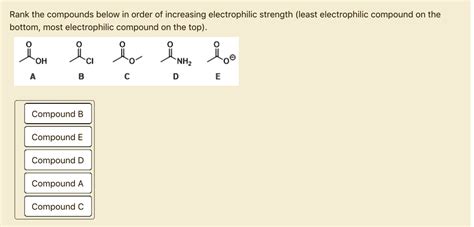
Table of Contents
Ranking Electrophilic Strength: A Comprehensive Guide
Electrophilicity, the tendency of an atom or molecule to attract electrons, is a crucial concept in organic chemistry. Understanding the relative electrophilicity of different structures is essential for predicting reaction pathways and outcomes. This article will delve into the factors influencing electrophilicity and rank various structures in order of decreasing electrophilic strength, providing a detailed explanation for each ranking. We'll explore carbocations, carbonyl compounds, halogens, and other important electrophilic species.
Factors Affecting Electrophilic Strength
Several factors contribute to the electrophilicity of a molecule or atom. The most significant include:
1. Positive Charge Density:
A positive charge directly implies a deficiency of electrons, making the species strongly electrophilic. The higher the positive charge density, the stronger the electrophile. For instance, a carbocation with a +2 charge will be more electrophilic than a carbocation with a +1 charge.
2. Electronegativity:
Atoms with high electronegativity attract electrons more strongly. Consequently, molecules containing highly electronegative atoms (like oxygen, fluorine, chlorine) can exhibit significant electrophilicity, even without a formal positive charge. The electronegative atom draws electron density from neighboring atoms, creating a region of partial positive charge that can attract nucleophiles.
3. Resonance Effects:
Resonance stabilization can significantly influence electrophilicity. If a positive charge can be delocalized through resonance, the charge density on any one atom is reduced, decreasing its electrophilicity. Conversely, if a positive charge is localized on a single atom, the electrophilicity increases.
4. Steric Hindrance:
Bulky substituents can hinder the approach of a nucleophile, decreasing the electrophilicity. Steric effects are particularly important in reactions involving bulky electrophiles and nucleophiles.
5. Inductive Effects:
Electron-withdrawing groups (EWGs) attached to an electrophilic center increase electrophilicity by pulling electron density away from the electrophilic center, making it more positive. Conversely, electron-donating groups (EDGs) decrease electrophilicity.
Ranking Electrophilic Structures
Let's now rank various types of electrophiles in order of decreasing electrophilic strength, considering the factors discussed above. This ranking is a generalization, and the relative electrophilicity can vary depending on the specific molecule and reaction conditions.
Note: This ranking is a general guideline, and the precise order can change depending on the specific substituents and reaction conditions.
1. Highly Stabilized Carbocations (e.g., allyl carbocation, benzyl carbocation): While possessing a positive charge, resonance delocalization significantly reduces the positive charge density, making them less electrophilic than primary or secondary carbocations. However, they are still considerably more electrophilic than most neutral molecules.
2. Primary Carbocations: These carbocations have only one alkyl group attached to the positively charged carbon. They are relatively unstable due to the concentrated positive charge and are therefore highly electrophilic.
3. Secondary Carbocations: With two alkyl groups, secondary carbocations are slightly more stable than primary carbocations due to hyperconjugation, thus slightly less electrophilic.
4. Tertiary Carbocations: Possessing three alkyl groups, tertiary carbocations are the most stable carbocations due to increased hyperconjugation and inductive effects. This increased stability reduces their electrophilicity compared to primary and secondary carbocations.
5. Acyl Halides (e.g., Acetyl Chloride): The carbonyl carbon in acyl halides carries a significant partial positive charge due to the electronegativity of the oxygen and the halogen. This makes them strong electrophiles, readily reacting with nucleophiles.
6. Acid Chlorides: Similar to acyl halides, acid chlorides exhibit strong electrophilicity due to the electron-withdrawing nature of the chlorine atom and the carbonyl group.
7. Acid Anhydrides: The carbonyl carbons in acid anhydrides have a partial positive charge, making them electrophilic, though less so than acyl halides due to resonance stabilization.
8. Aldehydes and Ketones: The carbonyl carbon in aldehydes and ketones possesses a partial positive charge, making them electrophilic. The electrophilicity of ketones is generally slightly lower than that of aldehydes due to the presence of two electron-donating alkyl groups.
9. Esters: Esters are less electrophilic than aldehydes and ketones due to the electron-donating effect of the alkoxy group (-OR). The carbonyl carbon still carries a partial positive charge, but resonance stabilization and the electron-donating group reduce its electrophilicity.
10. Carboxylic Acids: Carboxylic acids are even less electrophilic than esters due to the additional electron-donating hydroxyl group (-OH), which further stabilizes the carbonyl group.
11. Amides: Amides are the least electrophilic of the carbonyl compounds. The nitrogen atom donates electron density to the carbonyl group via resonance, significantly reducing the electrophilicity of the carbonyl carbon.
12. Halogens (e.g., Cl₂, Br₂): Halogens are electrophilic due to their high electronegativity, creating a region of partial positive charge on the halogen atom. However, their electrophilicity is generally lower than that of many carbonyl compounds. The electrophilicity decreases down the group (F₂ > Cl₂ > Br₂ > I₂).
13. Nitro Compounds: The nitro group (-NO₂) is a strong electron-withdrawing group, significantly increasing the electrophilicity of the carbon atom to which it is attached.
14. α,β-Unsaturated Carbonyl Compounds: The presence of a double bond conjugated to a carbonyl group leads to delocalization of the pi electrons, resulting in increased electrophilicity at the β-carbon. This is due to the resonance structures that place a positive charge on the β-carbon.
Conclusion
Understanding the factors influencing electrophilicity is crucial for predicting reaction mechanisms and outcomes in organic chemistry. While the above ranking provides a general guideline, the specific relative electrophilicity of different molecules can vary depending on the reaction conditions and specific substituents. Careful consideration of factors like positive charge density, electronegativity, resonance, steric effects, and inductive effects is essential for accurately assessing the electrophilicity of any given molecule. This comprehensive guide aims to equip you with a strong foundation to understand and predict reactivity in various organic chemical reactions. Further research into specific reaction mechanisms and the involved molecules will deepen your understanding of electrophilic behavior and reactivity. Remember to always consider the specific context of a reaction when assessing relative electrophilic strengths.
Latest Posts
Latest Posts
-
Ap Computer Science Principles 2021 Practice Exam Mcq
Apr 04, 2025
-
Graphing Skill 1 What Type Of Graph Is It
Apr 04, 2025
-
The Axial Skeleton Review Sheet 8
Apr 04, 2025
-
Lesson 4 Homework Practice Powers Of Monomials Answer Key
Apr 04, 2025
-
Which Is A False Statement Of Electronic Records
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Rank The Structures In Order Of Decreasing Electrophilic Strength . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
